Researchers at Australia’s Curtin University, in conjunction with scientists from Shanghai University, Jilin University, Chinese Academy of Sciences, Fudan University and TCL Research, have developed an exciting new method to create blue quantum dot LEDs without using toxic heavy metals such as cadmium. Quantum dots, which are tiny semiconductor particles that emit bright, highly precise colors, have long been seen as the next step in display technology. The problem, however, has always been how to produce a stable and efficient pure-blue device without relying on cadmium, which poses environmental and health concerns.
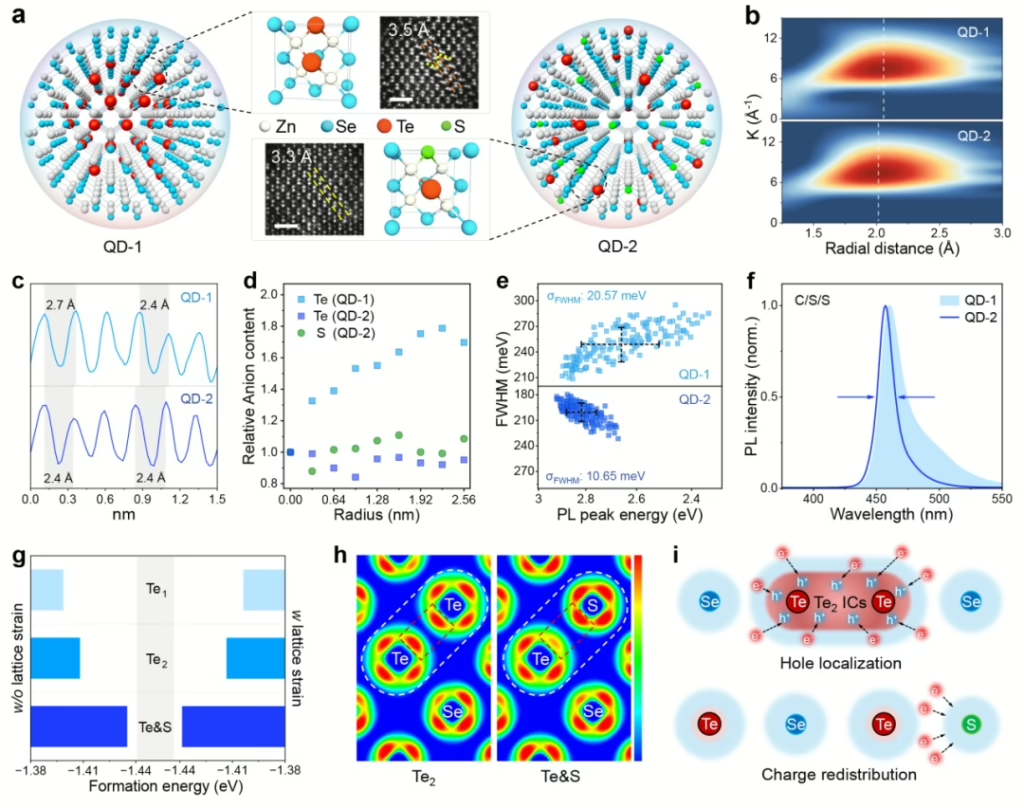
In zinc selenide–telluride quantum dots, tellurium atoms tend to cluster because they have a natural propensity to bond with each other (forming Te–Te bonds, sometimes referred to as polytelluride species). This tendency can be exacerbated by the relative difficulty of uniformly incorporating tellurium into the zinc selenide lattice—tellurium has a larger atomic radius and slightly different bonding characteristics than selenium. If growth conditions are not precisely controlled, tellurium-rich regions can form more readily than uniformly distributed tellurium atoms, leading to the formation of these aggregated “pockets” of Te. Those pockets, in turn, introduce defects and compositional inhomogeneities in the quantum dots that degrade their optical and electrical properties.

The research team tackled this challenge by fine-tuning the atomic structure of zinc-based quantum dots in a way that prevents defects known to reduce brightness and overall device stability. They introduced sulfur, which helped prevent the clumping of tellurium and improved the dots’ uniformity. By ensuring the materials in the quantum dots were carefully balanced, they could produce a pure-blue color with minimal unwanted emission and prolong the device’s lifespan.
This approach led to a blue quantum dot LED with an external quantum efficiency of 24.7%—on par with or even surpassing traditional cadmium-based designs, and a lifetime of nearly 30,000 hours at practical brightness levels. The result is an eco-friendly display technology that may pave the way toward safer TVs, smartphones, and other electronic displays.
Reference
Wu, Q., Cao, F., Yu, W. et al. Homogeneous ZnSeTeS quantum dots for efficient and stable pure-blue LEDs. Nature (2025). https://doi.org/10.1038/s41586-025-08645-4

