Researchers at Lehigh University in Bethlehem, Pennsylvania, are looking into improved production methods for nanoparticles. Quantum dots (QDs) are the nanoparticle of greatest interest to the display industry but they are certainly not the only nanoparticles of interest to other industries.

They have a wide array of applications in fields ranging from diagnostic imaging to solar cells. Unfortunately the conventional syntheses of these nanoparticles occur under harsh conditions in toxic organic solvents. For this reason, the bacterial synthesis of nanoparticles has received considerable attention for its economic and environmental advantages over conventional synthesis.
Nanoparticles produced by conventional methods are also expensive. Quantum dots cost on the order of $1000 – $10,000 per gram. The researchers believe that this cost could be brought down by a factor of as much as 10X using biological production techniques instead of traditional solvent chemistry techniques.
 Undergraduate Robert Dunleavy and Professor Bryan Berger, Associate Professor of Chemical and Biomolecular Engineering at Lehigh University
Undergraduate Robert Dunleavy and Professor Bryan Berger, Associate Professor of Chemical and Biomolecular Engineering at Lehigh University
As an undergraduate research project, Robert Dunleavy, working with Professor Bryan Berger, has synthesized Cadmium Sulfide (CdS) and Lead Sulfide (PbS) quantum dots using two different biological techniques. Many quantum dots used in displays today are CdS, although the display industry is also supplying and developing cadmium-free quantum dots because of the high toxicity of cadmium. Japan banned cadmium-based green phosphors from CRTs in the early 1970s and by the time I started to work in the CRT industry in 1979, all CRT phosphors were cadmium-free. Of course, a CRT could contain grams of cadmium but QD-based LED TVs are more likely to milligrams of cadmium, a far smaller hazard. (QDs with cadmium are reported to be completely unacceptable to Apple and Samsung – Man. Ed.)
The researchers at Lehigh first used the bacteria Stenotrophomonas maltophilia (Strain SMCD1) in a solution of growth media containing Cd+2 and L-cysteine, a sulphur-containing chemical. In response, the bacteria synthesized CdS QDs. The size of the quantum dots and therefore the wavelength continuously grew over the 30 minute – 360 minute times of the experiments, resulting in quantum dots with a variety of colors, as shown in the figure.

The researchers couldn’t stop here. S. Maltophilia is a highly infectious bacteria, sometimes called a “superbug”, that had been brought to Dr. Berger’s attention by medical people because it was 1) antibiotic resistant and 2) able to survive in metallic environments that would be toxic to most bacteria. Clearly a bacteria like this could not be used in any large-scale industrial production process.
The researchers investigated exactly what it was in the SMCD1 bacteria that produced the quantum dots. After investigating a variety of proteins using protein mass spectrometry, they focused their attention on the enzyme cystathionine ?-lyase (CSE), which catalyzes the formation of pyruvate, H2S and ammonia from L-cysteine. As L-cysteine was required for CdS QD synthesis using SMCD1, CSE appeared likely to be responsible for the enzymatic synthesis of CdS QDs.
The gene in S. maltophilia that coded for CSE was synthesized and cloned. This gene was then used to produce relatively large quantities of recombinant putative cystathionine ?-lyase (smCSE) for use in further experiments. They found that high quality cadmium sulfide and lead sulfide nanoparticles were produced upon incubation of CSE with cysteine and cadmium or lead salts, respectively. Without CSE, these solutions are stable and no material of any sort precipitates out. The researchers say smCSE mineralizes CdS from an aqueous cadmium acetate solution by catalytic production of reactive H2S from L-cysteine.
They also say smCSE controls nanocrystal growth within the quantum confined size range. This function of enzymatic nanocrystal templating was demonstrated by substituting reactive Na2S as the sulfur source. In this solution, the catalytic function of amCSE is not needed because the materials are already reactive. In the absence of smCSE, this solution will produce bulk CdS. With the same solutions, nanocrystal formation is observed when smCSE is present to control the growth.
Thus, the smCSE actually serves dual purposes in the process of forming quantum dots. First it catalyzes the L-cysteine into required sub-components and second it serves as a template for the formation of nanoparticles rather than bulk CdS material. In terms of industrial production of QDs, an enzyme that can be synthesized in nearly unlimited quantity is far preferable to a dangerous and highly contagious bacteria.
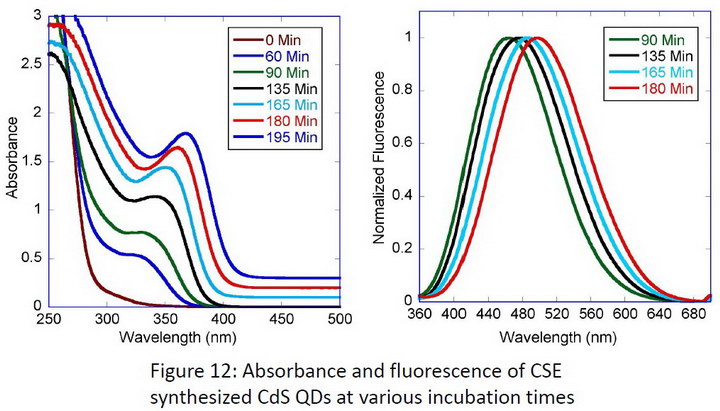
The optical properties of QDs produced by CSE are shown in Figure 12. Note that in this figure, the absorbance peak is at about 360nm. Theory and past research says this absorbance wavelength corresponds to a quantum dot of approximately 2nm in diameter. The nanoparticles produced were also imaged with a high-resolution transmission electron microscope (HRTEM). These HRTEM images show well-dispersed crystalline CdS nanoparticles with diameters of approximately 2 to 3 nm after a 2 hour growth time, which is in agreement with the expected QD size from absorbance spectroscopy.

Of course, an undergraduate research project is a long way from commercialization of a process. Future work identified by the Lehigh researchers involves improving the quality of the synthesized nanoparticles and investigating on a mechanistic level why cystathionine ?-lyase can produce such high quality particles. Still, according to the researchers, “This dual-function, single-enzyme, aerobic, and aqueous route to functional material synthesis demonstrates the powerful potential of engineered functional material biomineralization.”
Results of this research has been published as “Single-enzyme biomineralization of cadmium sulfide nanocrystals with controlled optical properties” by Robert Dunleavy, Li Lu, Christopher J. Kiely, Steven McIntosh and Bryan W. Berger in the Proceedings of the National Academy of Sciences of the United States of America, May 10, 2016. (http://www.pnas.org/content/113/19/5275 On-line publication had been in April.) A less technical version of this research was published in the New York Times on May 5th. –Matthew Brennesholtz

